Косметика всегда играла важную роль в жизни людей, будь то дело о красоте, уходе
В нашей статье мы поговорим о том, какие инструменты наиболее важны для работы парикмахера,
Переплет документов, таких как дипломы, курсовые и диссертации, является важным этапом в подготовке этих
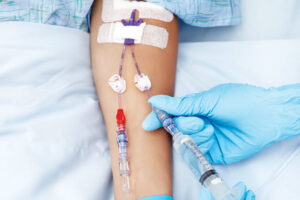
Капельница от наркотиков действенное лечение на дому Употребление наркотиков приводит к постепенному возникновению зависимости.
Детские новогодние подарки сладкие в Москве - самые популярные сюрпризы для малышей. Разнообразные наборы
Кожаные кошельки - это не просто аксессуар для хранения денег, это небольшое произведение искусства,
Покупка золотых обручальных колец в онлайн-магазине может предоставить множество преимуществ, таких как больший выбор,
Hair-Man - это сайт, посвященный стильным мужским стрижкам. На этом сайте вы можете найти
Создание выпускных альбомов - это увлекательный и творческий процесс, который запечатлевает важные моменты и
Брезент купить можно в многих специализированных магазинах для строительства, автомобильных магазинах, интернет-магазинах и других
Теплоизоляционные материалы - это материалы, предназначенные для улучшения теплозащиты и энергоэффективности зданий.
Converse – это обувь, которая подходит к любому стилю. Белые короткие кеды – это
Современные технологии пластической хирургии предлагают широкий спектр процедур для того, чтобы помочь вам достичь
Для пошива верхней одежды используют различные виды тканей, каждый из которых имеет свои преимущества
Особенности черных ресниц для наращивания могут незначительно отличаться в зависимости от производителя и вида
Коллаген порошок - это продукт, полученный из природных источников коллагена, таких как животное сырье
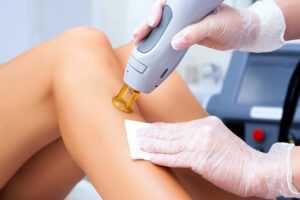
Лазерное удаление волос — аппаратная процедура, которая проводится в условиях клиники с помощью дорогостоящего
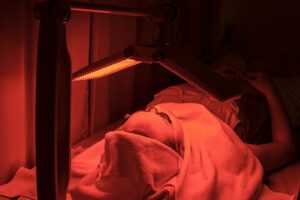
На заметку. Практически доказана роль иммунологического компонента при фотодинамической терапии, что говорит о перспективности
Следуя этим простым рекомендациям, вы сможете выбрать действительно хороший подарок. Если у вас возникли
После разговора с госпитализированным пациентом врач решает, какое лечение использовать. Если пациент согласится, врач
Профессия ногтевого стилиста становится все более требовательной. Умения красиво накрасить ногти или нанести гель
Описание и сравнение домашних и салонных способов выпрямления волос. С помощью фена, утюжка, крахмальное,
ТОП лучших детских причесок для девочек на каждый день или торжественный праздник. Описание, как
Торговля на финансовых рынках сейчас осуществляется онлайн с помощью специальных программ – торговых терминалов.
Перед написанием бизнес-плана специалистами или владельцем компании определяются задачи, средства по их выполнению. Разработанный
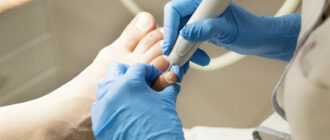
Специалист клиники Remedy Lab Эльмира Мингалеева — об особенностях подологического педикюра и средствах для
И опять мы заводим разговор про краевые показатели. Все симптомы, такие как болевые ощущения,
Квадратное сечение таких труб позволяет равномерно распределять все нагрузки на изгиб, что дает возможность
Еще один вопрос, который волнует невест – это какой цвет короткого свадебного платья выбрать.